Unwittingly, chemists have tied the smallest and tightest knot ever, beating the previous Guinness World Record – this amazing feat consists of just 54 atoms, which have been tied together three times to form a cloverleaf knot. The shape, which resembles a three-leaf clover, has no loose ends, is the simplest way to form a trivial knot, and is a fundamental element of the mathematical theory of knots.
Four years ago, in 2020, Chinese chemists made a chain of 69 atoms that intersected three times to create the same shape, but researchers from the University of Western Ontario in Canada and the Chinese Academy of Sciences came together to beat the record. As the ratio of atoms to transitions decreases, the strength of the molecular knot increases. The 2020 node had a transit ratio (RC) of 23, the last one being 18.
Molecular knot and science
Most organic molecular knots have an RC between 27 and 33, but experts don't know how small or tight they can still be made. Some quantum chemistry calculations suggest that the most stable trilobite structure would be around 50 molecules, which means we are very close to the theoretical limit.
With this achievement, scientists are closer than ever to the microscopic knots that naturally form in DNA, RNA, and many other proteins in our bodies. Plastics and polymers could also be manufactured in more efficient ways based on the discovery, as their properties depend on the size of the complex structure, according to the researchers.
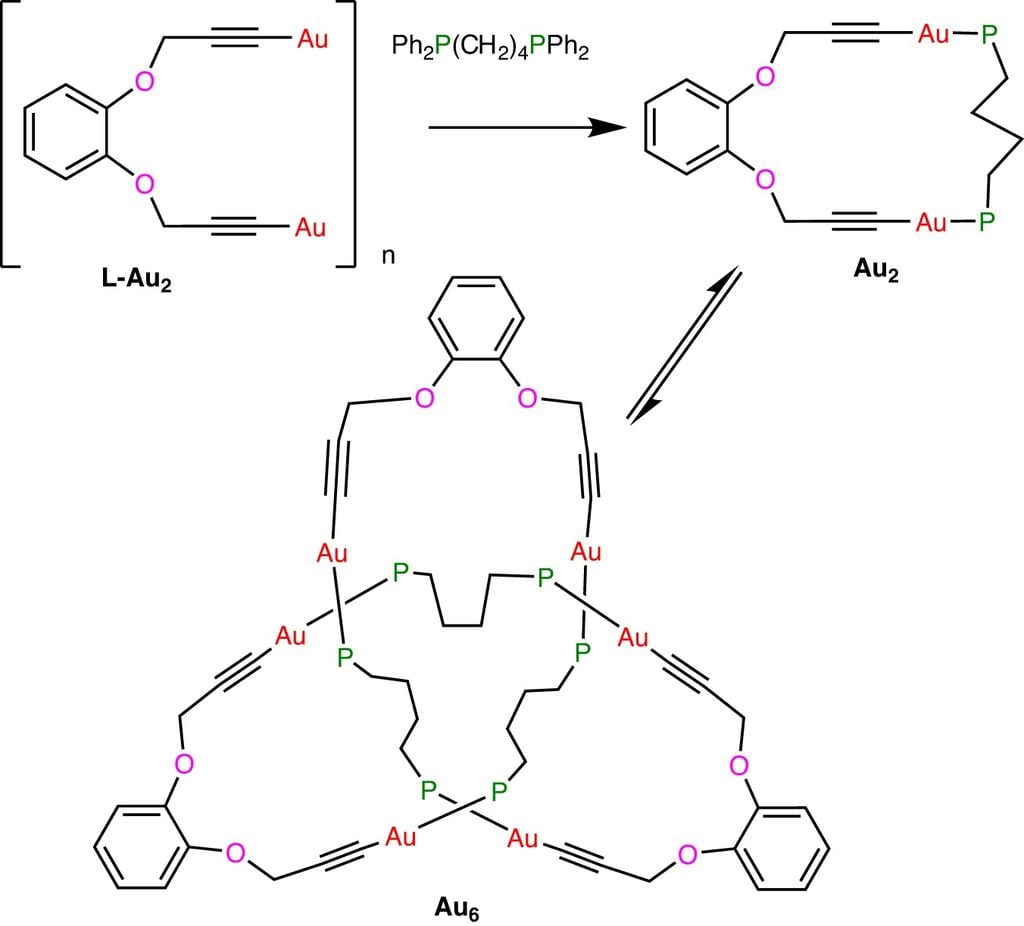
To the site News worldChemist Richard Budivat told how this happened by chance. He and his colleagues worked in the laboratory with metal acetylides — which are alkenes, a type of hydrocarbon — with the hydrogen removed from their end.
The product is very useful in chemical reactions carried out by scientists. When they linked the gold acetylide to another carbon structure called a diphosphine bond, the team ended up creating a cloverleaf knot instead of a gold chain (or catenane). Creating molecular knots has been attempted by chemists by directing helical chains into desired structures via metal ions since 1989.
The 2020 knot is made by bending and interlocking the molecular chain, and when the metal atoms are removed at the end of the process, the knot can no longer be undone. The new golden knot is different, because it curls on its own. It's a complex system that scientists admit they don't know how it works, but they say they're excited to create similar systems in the future.
source: nature, Nature Communications With information from News world

“Coffee trailblazer. Social media ninja. Unapologetic web guru. Friendly music fan. Alcohol fanatic.”

